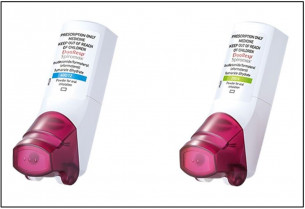
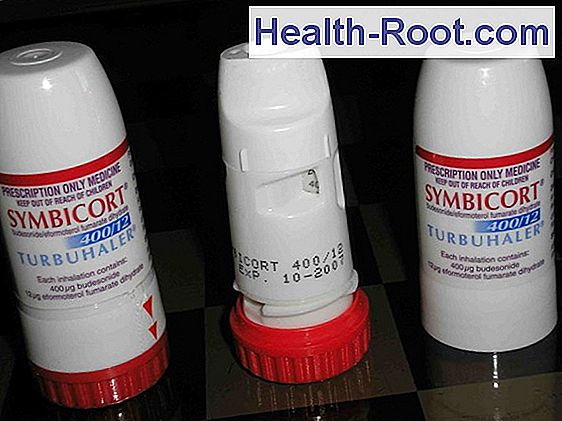
PDF) Assessment of inhalation errors, training time and patient preference for DuoResp® Spiromax® and Symbicort® Turbuhaler® in patients with asthma and COPD

Teva recibe la autorización de comercialización en la UE para Seffalair Spiromax y BroPair Spiromax para el tratamiento del asma

Spiromax device. Copyright of Teva UK Limited. Reproduced with permission. | Download Scientific Diagram

Teva receives EU Marketing Authorisation for Seffalair® Spiromax® and BroPair® Spiromax® (salmeterol xinafoate / fluticasone propionate) | Business Wire
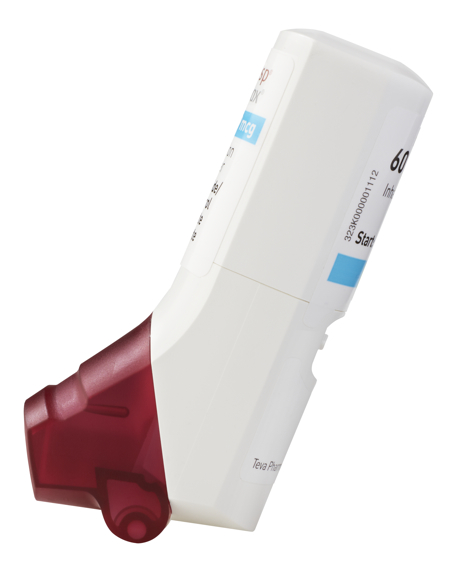
DuoResp Spiromax 320micrograms/dose / 9micrograms/dose dry powder inhaler ( Teva UK Ltd) 60 dose - RightBreathe

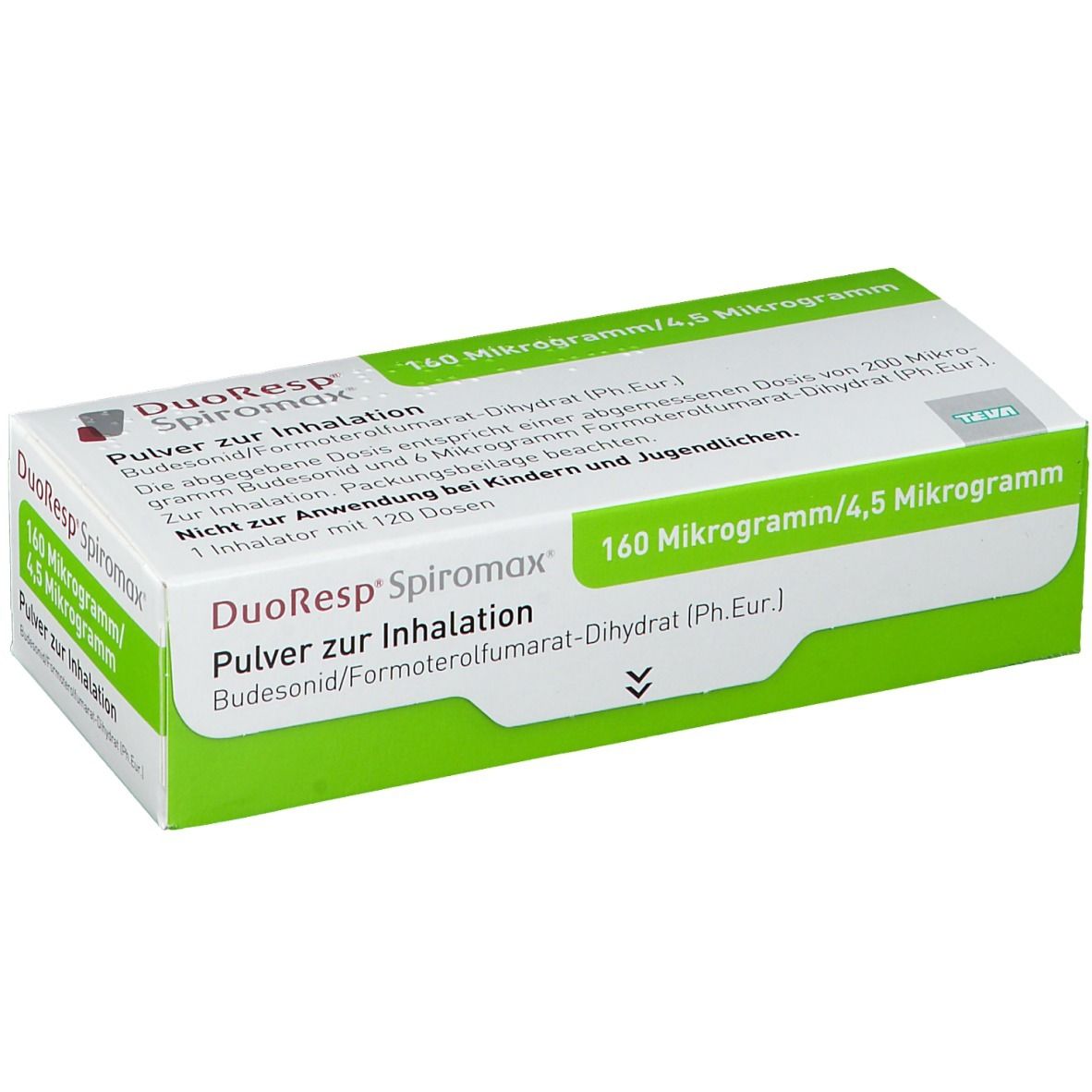
DuoResp Spiromax (ДуоРесп Спиромакс) 160mkg/4,5mkg, 1 ингалятор 120 доз,Teva Pharmaceutical Industries Ltd., Израиль | ИНТЕРНЕТ-АПТЕКА liki36-6.com