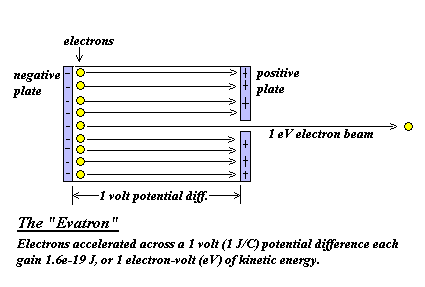
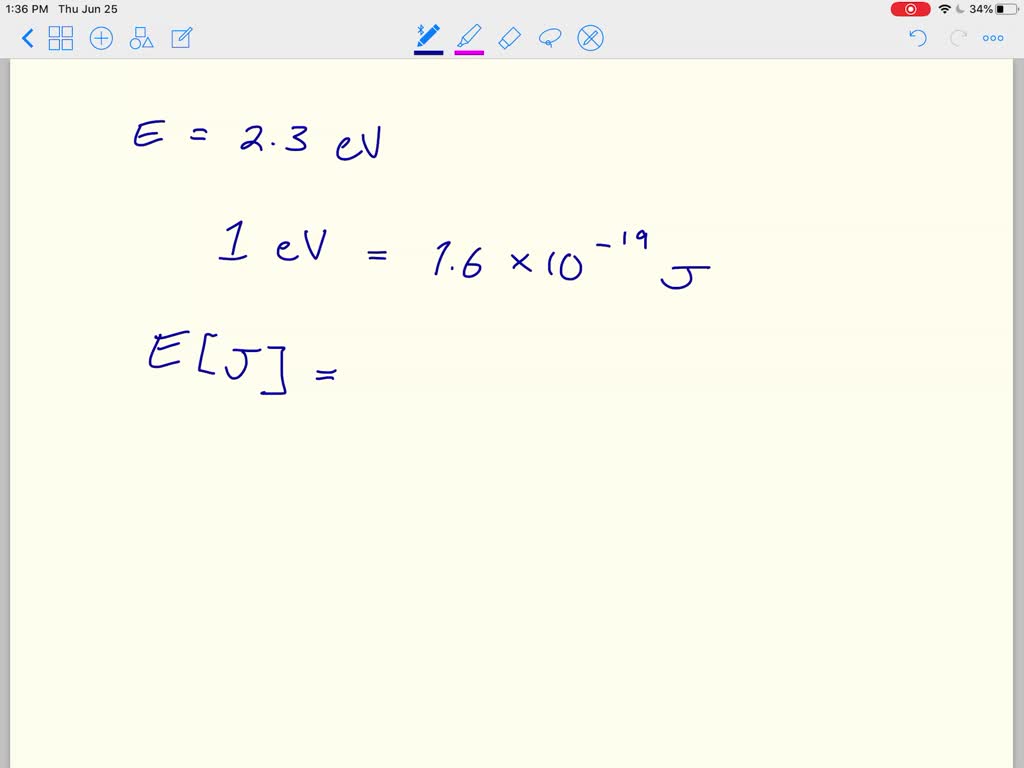SOLVED:The electron volt (eV) is a convenient unit of energy for expressing atomic-scale energies. It is the amount of energy that an electron gains when subjected to a potential of 1 volt;

Chapter 5. Thermonuclear Fusion 1.Introduction 2.Thermonuclear Reactions and Energy Production 3.Fusion in a Hot Medium 4.Progress Towards Fusion Power. - ppt download

The ionization enthalpy of hydrogen is 1312 0 kilo joule per mole Express the value in electron volt per - Chemistry - Classification of Elements and Periodicity in Properties - 11781691 | Meritnation.com

SOLVED:Using the Planck energy calculate the energy of the Balmer Series Alpha-Line, which has a wavelength in vacuum of Ao = 656.28 nm Express your answer in electron volts (eV): Note that