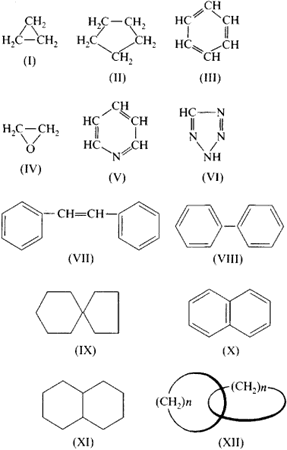
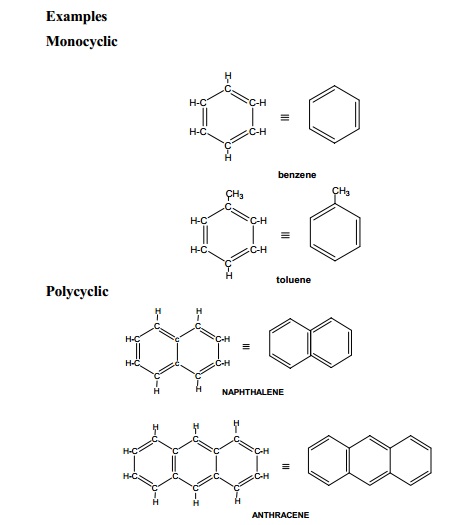
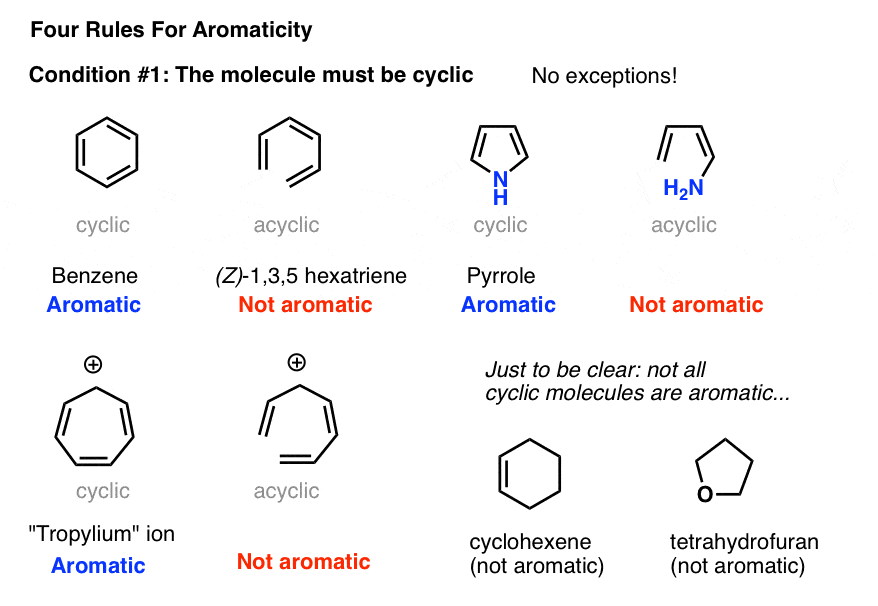
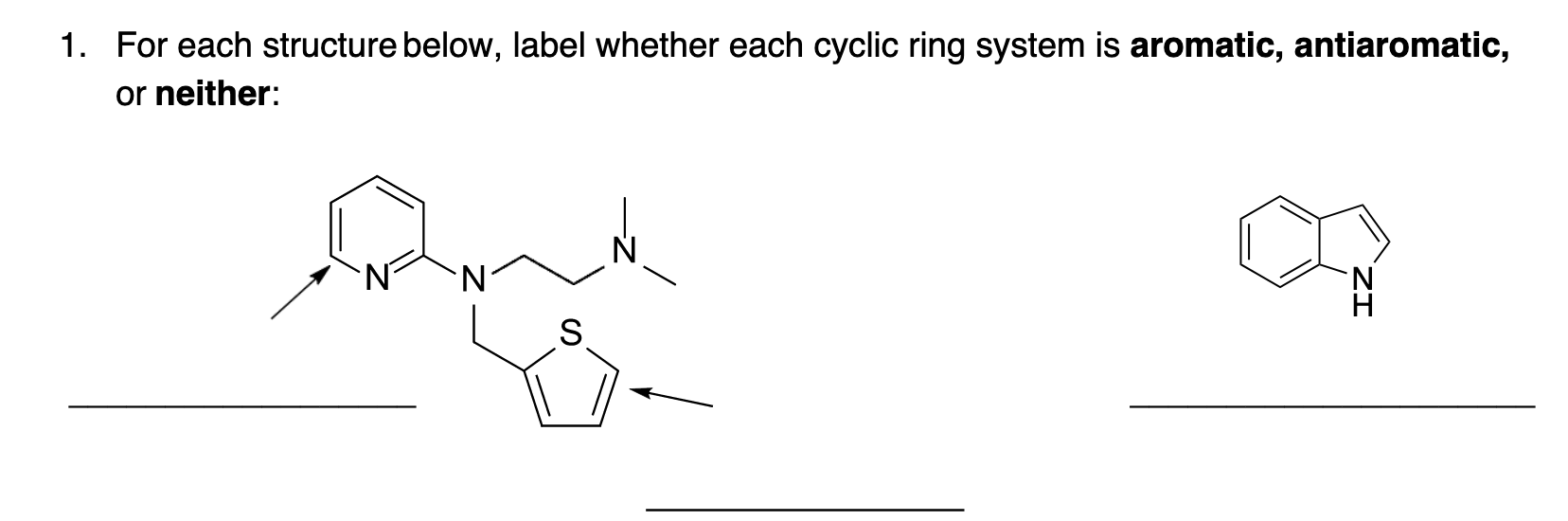
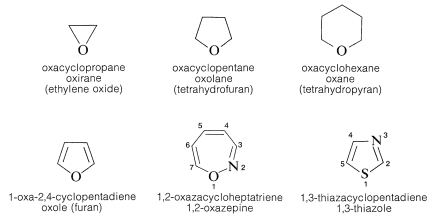
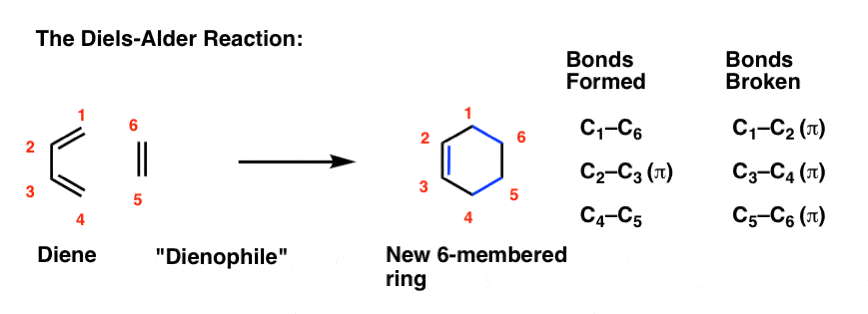
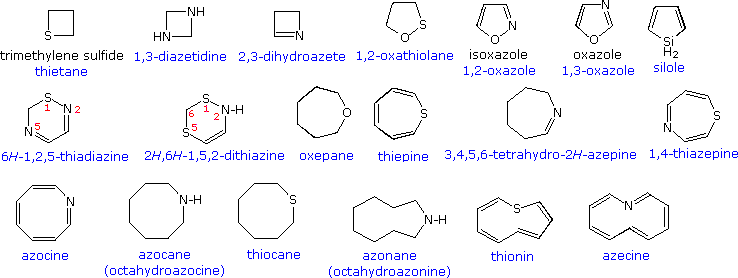
Utilizing IUPAC rules, name each of the following cyclic compounds. Use cis and trans where appropriate . See atachment | Study.com
![Ring expansion of cyclic 1,2-diols to form medium sized rings via ruthenium catalyzed transfer hydrogenative [4+2] cycloaddition - Chemical Communications (RSC Publishing) Ring expansion of cyclic 1,2-diols to form medium sized rings via ruthenium catalyzed transfer hydrogenative [4+2] cycloaddition - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C4CC03983A)
Ring expansion of cyclic 1,2-diols to form medium sized rings via ruthenium catalyzed transfer hydrogenative [4+2] cycloaddition - Chemical Communications (RSC Publishing)

Naphthenic acyclic (Z = 0) and single ring cyclic (Z = -2) acids of... | Download Scientific Diagram

Scheme 1. Enzymatic ring opening polymerization (eROP) of cyclic esters... | Download Scientific Diagram

From Cyclic Ketimines to α‐Substituted Cyclic Amino Acids and their Derivatives: Influence of Ring Size and Substituents on Stability and Reactivity of Cyclic Aminonitriles - Voznesenskaia - 2019 - European Journal of
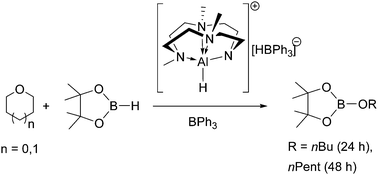
Ring-opening of cyclic ethers by aluminum hydridotriphenylborate - Chemical Communications (RSC Publishing)
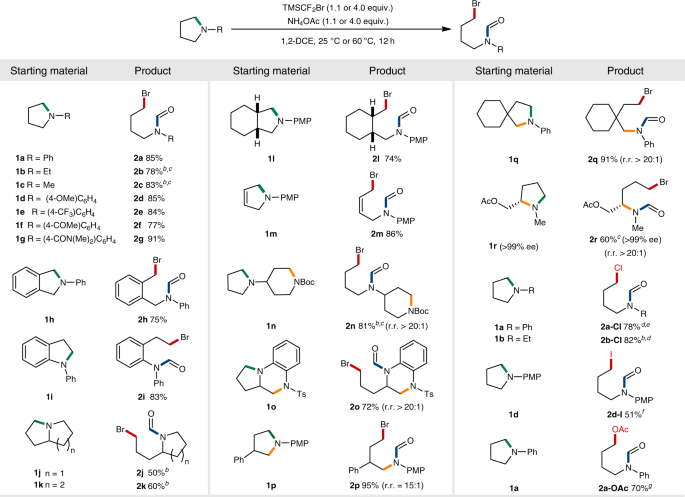
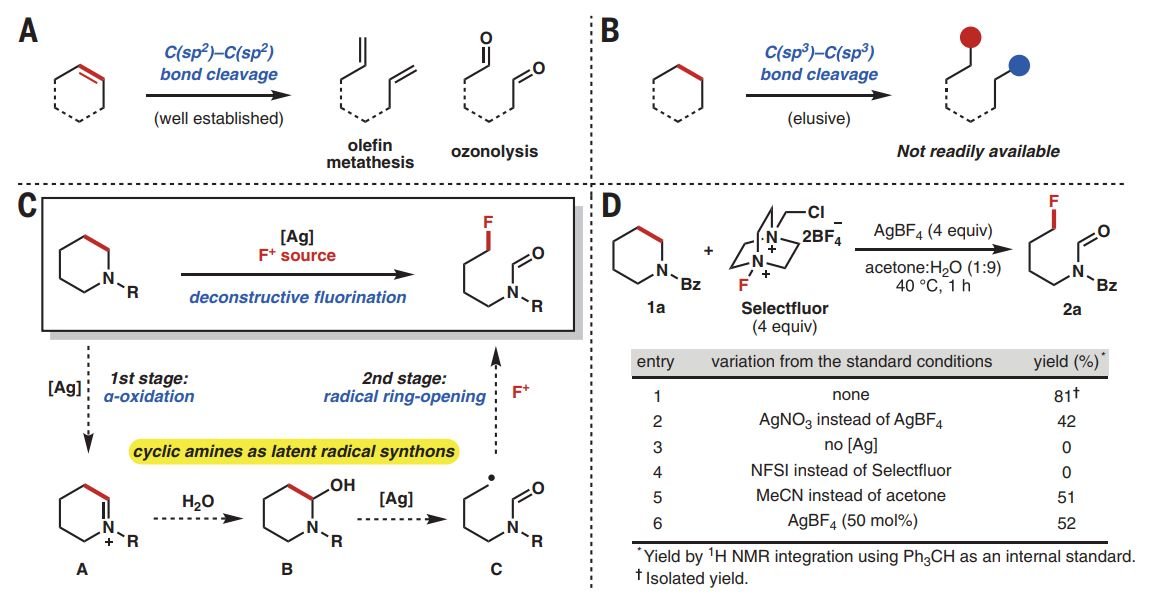
Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications
Patterns of ring closure for 3-to 6-membered rings (endo-tet processes... | Download Scientific Diagram