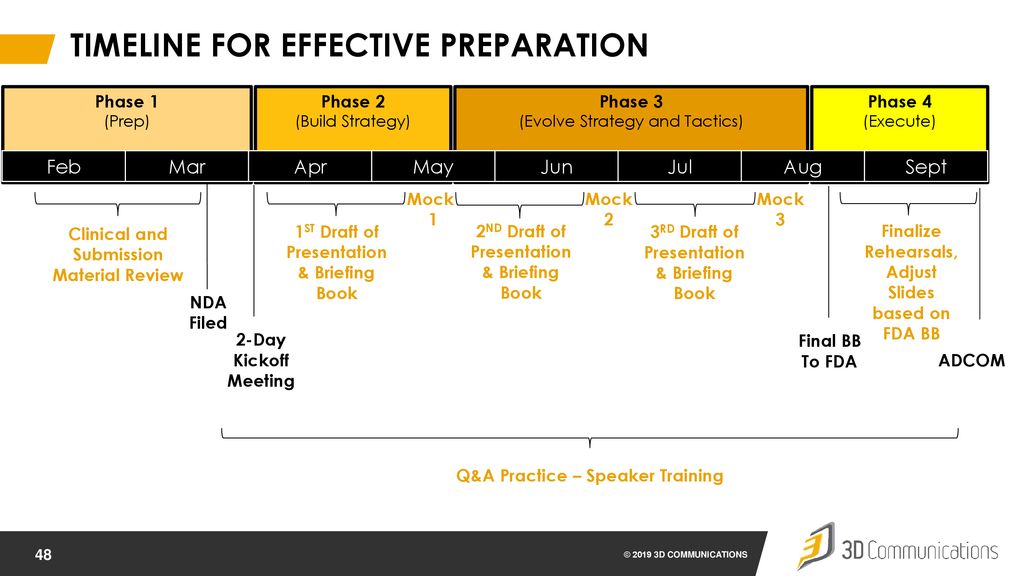
Meg Tirrell on Twitter: "Proposed timing for Pfizer booster shot in FDA briefing documents is 6 months out from primary series - not 8, which was what US health officials suggested https://t.co/U2tCyCOfdQ" /

Fillable Online fda FDA Briefing Information for the November 17, 2011 Meeting of the ... - fda Fax Email Print - pdfFiller
![FDA Briefing Document - Informa /media/Supporting...¢ discussed below, and in more detail in the Cross¢â‚¬¯Disciplinary - [PDF Document] FDA Briefing Document - Informa /media/Supporting...¢ discussed below, and in more detail in the Cross¢â‚¬¯Disciplinary - [PDF Document]](https://cdn.vdocument.in/img/1200x630/reader022/reader/2020052010/5e4a738fd892562c6c545653/r-1.jpg?t=1645315921)
FDA Briefing Document - Informa /media/Supporting...¢ discussed below, and in more detail in the Cross¢â‚¬¯Disciplinary - [PDF Document]
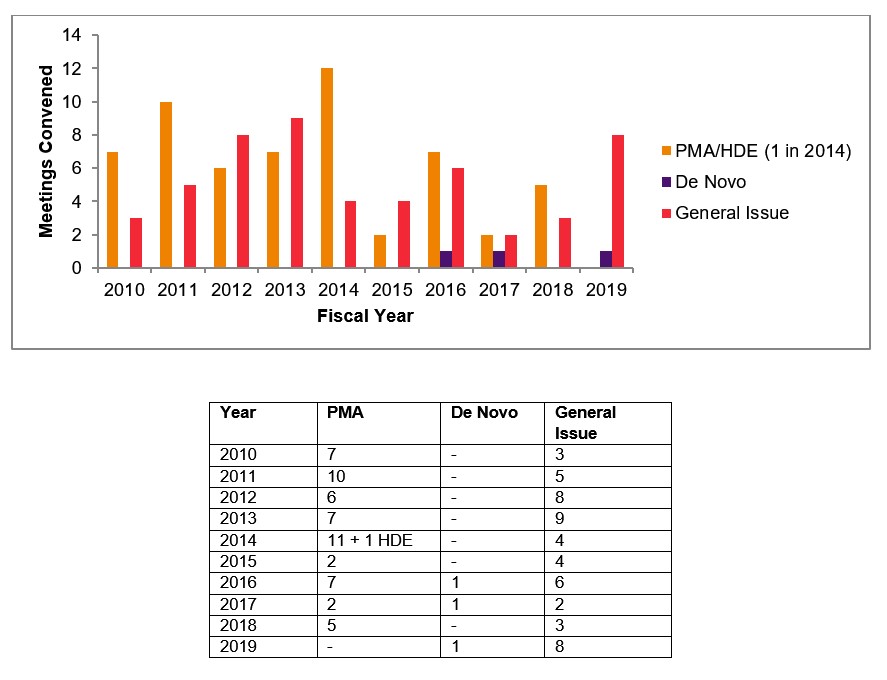
Bad News for Device Sponsors: Panel Meetings were Already Going the Way of the Homework Assignment, and COVID Might “Put the Nail in the Coffin” - Food and Drug Law Institute (FDLI)

FDA advisory committee recommends not approving FibroGen's anemia drug candidate roxadustat - Drug Discovery and Development

FDA advisory committee rejects recommending approval of lower-dose empagliflozin for type 1 diabetes

FDA Posts Briefing Documents for Advisory Meeting Reviewing sNDA for EXPAREL® as a Nerve Block for Regional Analgesia | Pacira BioSciences, Inc.