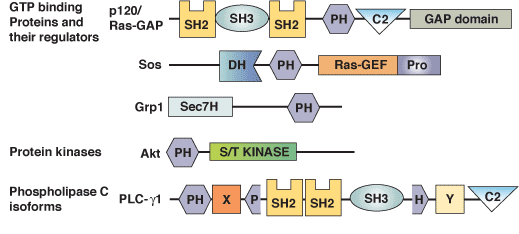
p120Ras-GAP binds the DLC1 Rho-GAP tumor suppressor protein and inhibits its RhoA GTPase and growth-suppressing activities | Oncogene

Structure of the GAP domain of LepB. (A) Domain architecture of LepB... | Download Scientific Diagram

The Ras GTPase-activating Protein (GAP) Is an SH3 Domain-binding Protein and Substrate for the Src-related Tyrosine Kinase, Hck ∗ - Journal of Biological Chemistry

Google Bridges Domain Gaps in Semantic Segmentation of LiDAR Point Clouds | by Synced | SyncedReview | Medium

ELMO Domains, Evolutionary and Functional Characterization of a Novel GTPase-activating Protein (GAP) Domain for Arf Protein Family GTPases* - Journal of Biological Chemistry
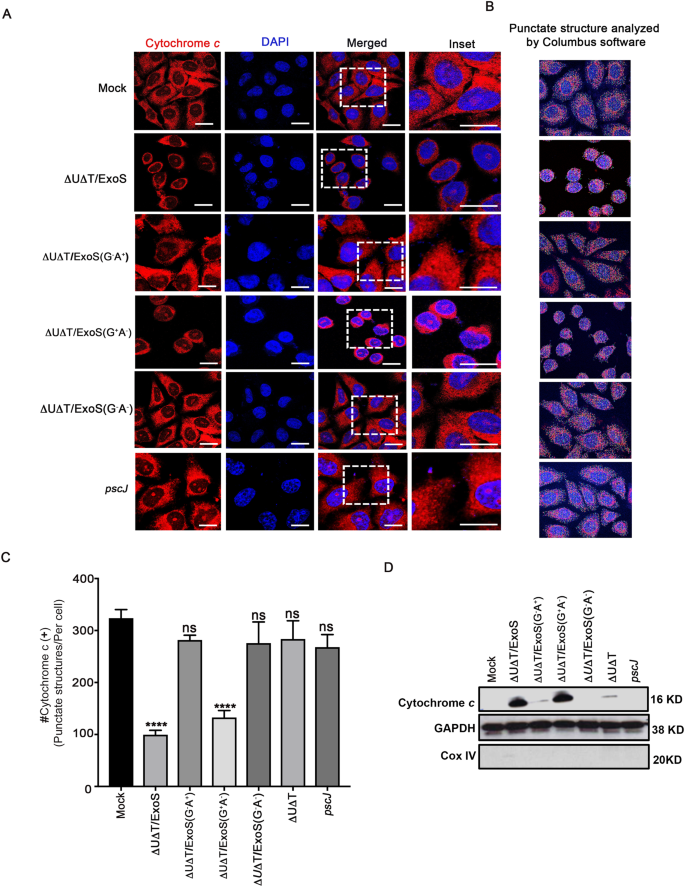
Pseudomonas aeruginosa ExoS Induces Intrinsic Apoptosis in Target Host Cells in a Manner That is Dependent on its GAP Domain Activity | Scientific Reports
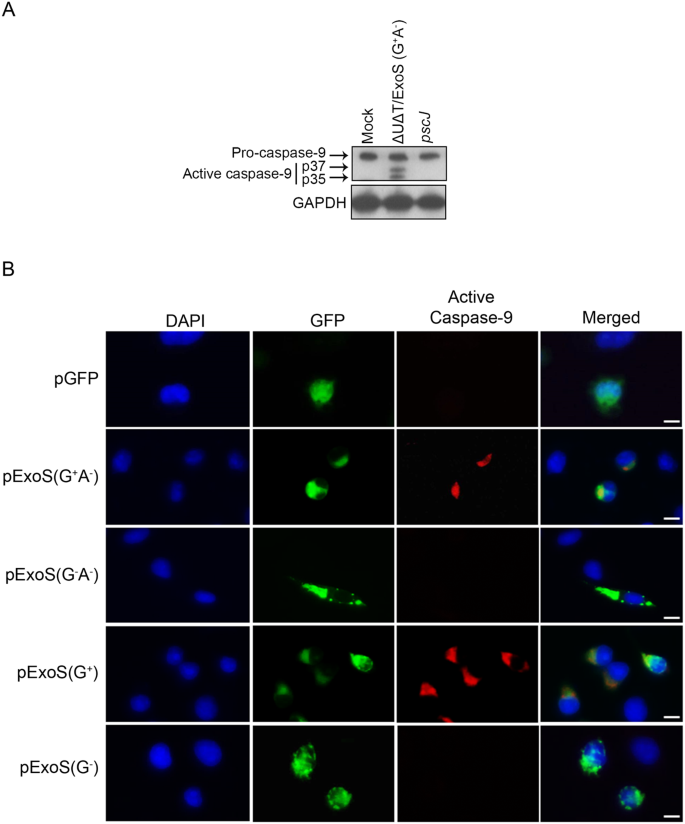
Pseudomonas aeruginosa ExoS Induces Intrinsic Apoptosis in Target Host Cells in a Manner That is Dependent on its GAP Domain Activity | Scientific Reports
GitHub - Omkar-Ranadive/Domain-Adaptation-CycleGAN: Closing the domain gap between simulator generated images and real-world images using CycleGAN

The BNIP-2 and Cdc42GAP Homology (BCH) Domain of p50RhoGAP/Cdc42GAP Sequesters RhoA from Inactivation by the Adjacent GTPase-activating Protein Domain | Molecular Biology of the Cell

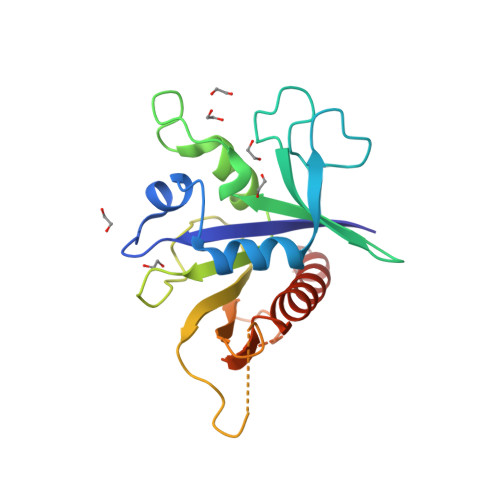
Structure of the TSC2 GAP Domain: Mechanistic Insight into Catalysis and Pathogenic Mutations.,Structure - X-MOL

![PDF] Bridging the Day and Night Domain Gap for Semantic Segmentation | Semantic Scholar PDF] Bridging the Day and Night Domain Gap for Semantic Segmentation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b9e3ceef50f799b371396652e31e0d3b09611500/1-Figure1-1.png)