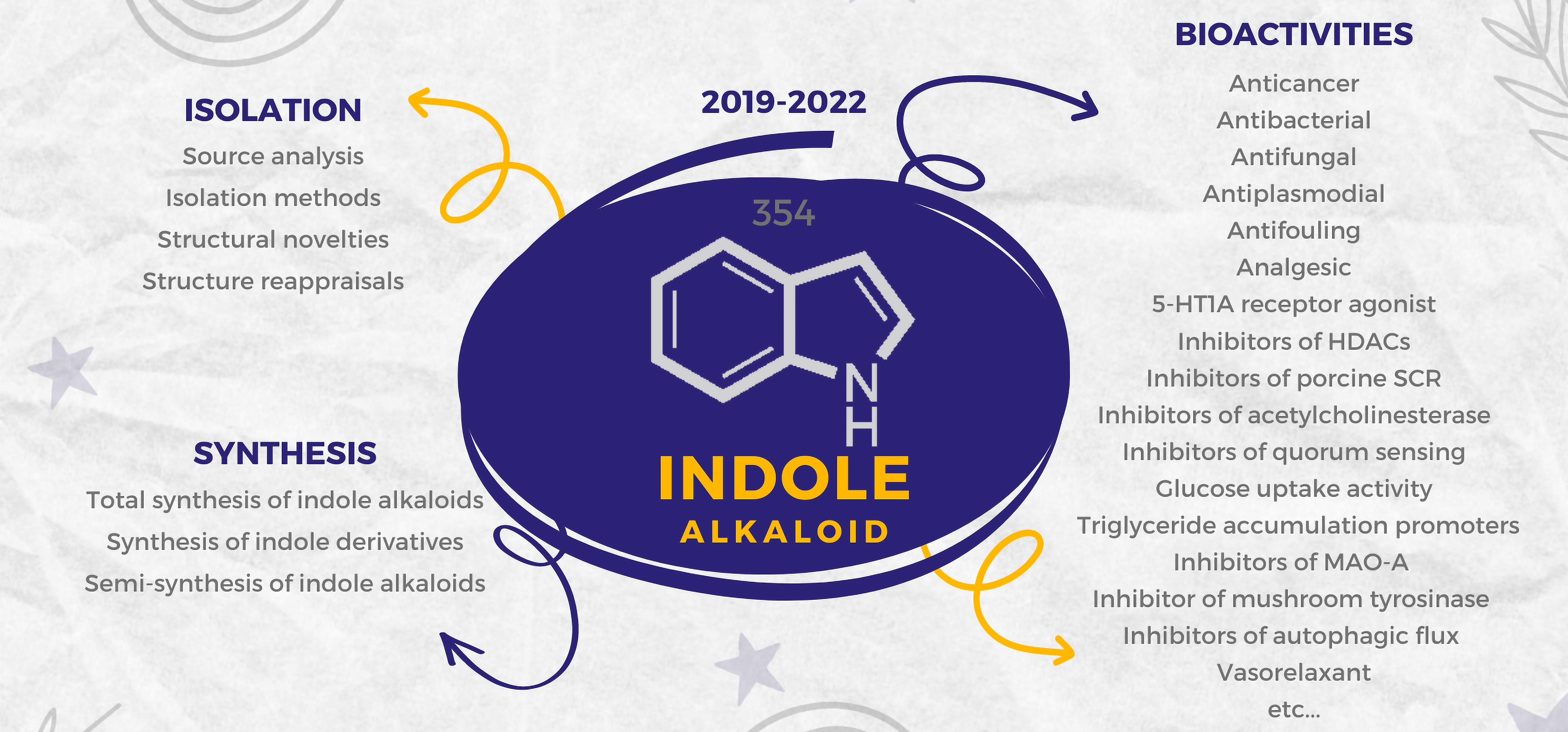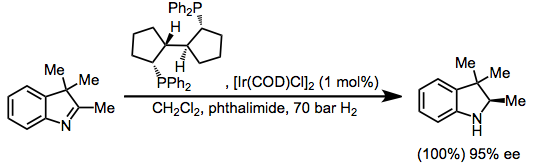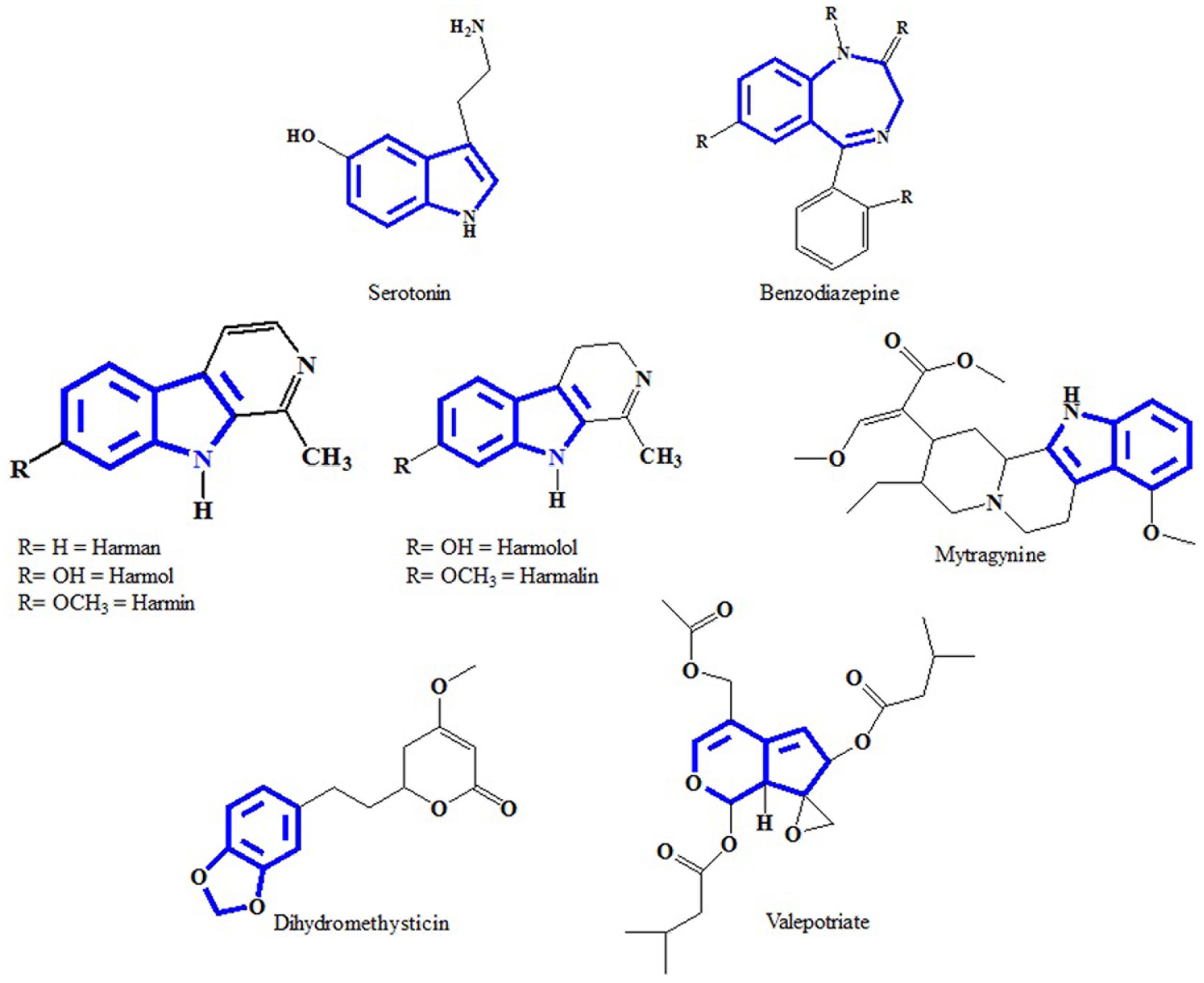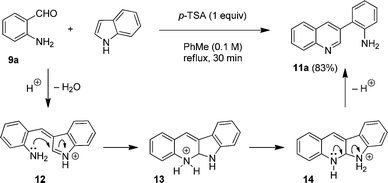
Divergent reactions of indoles with aminobenzaldehydes: indole ring-opening vs. annulation and facile synthesis of neocryptolepine - Chemical Science (RSC Publishing) DOI:10.1039/C1SC00506E

Au(I)-Catalyzed Pictet–Spengler Reactions All around the Indole Ring | The Journal of Organic Chemistry

A) Structure of the perdeuterated indole ring-d 6 . The molecule is... | Download Scientific Diagram
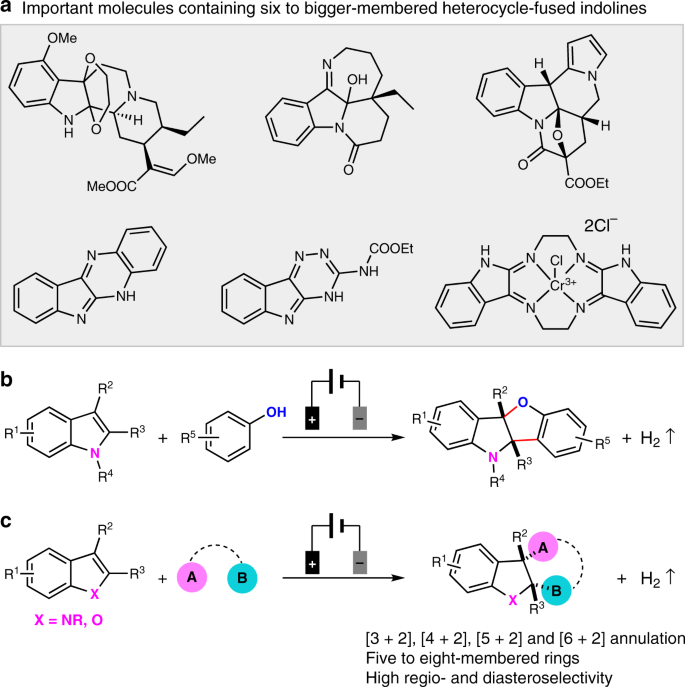
Electrooxidation enables highly regioselective dearomative annulation of indole and benzofuran derivatives | Nature Communications

Two-Carbon Ring Expansion of Cyclobutanols to Cyclohexenones Enabled by Indole Radical Cation Intermediate: Development and Application to a Total Synthesis of Uleine | ACS Catalysis

Synthesis of Structurally Diverse 2,3-Fused Indoles via Microwave-Assisted AgSbF6-Catalysed Intramolecular Difunctionalization of o-Alkynylanilines | Scientific Reports

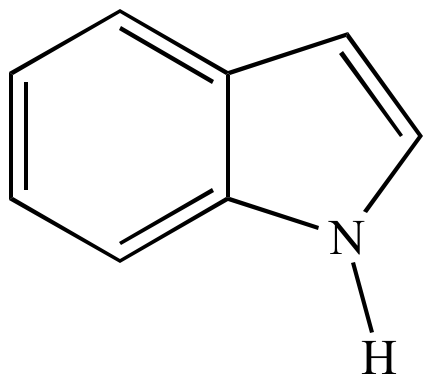
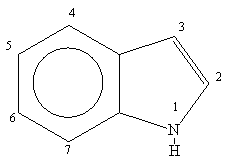
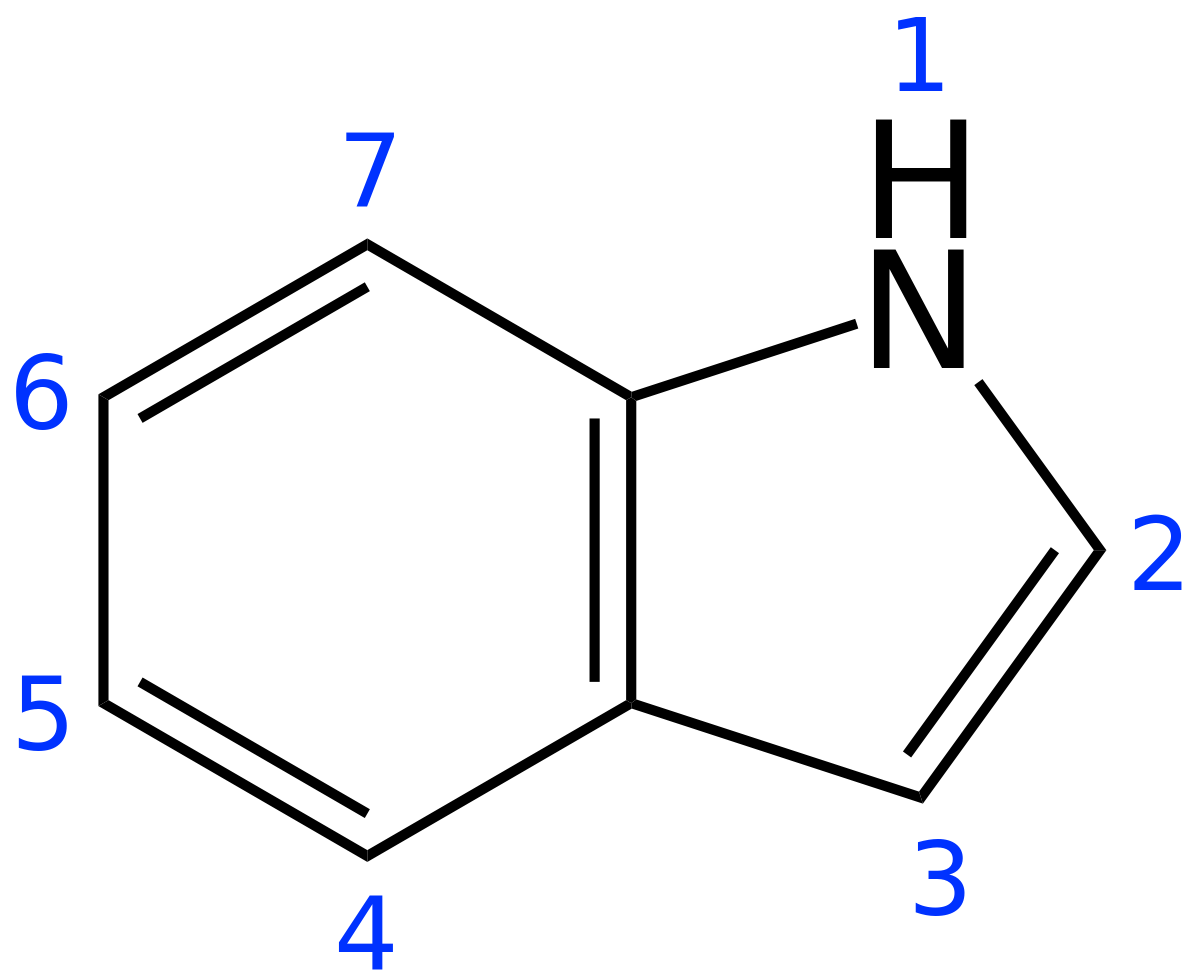
Indole is an aromatic heterocycle that has a benzene ring fused to a pyrrole ring. Draw an orbital picture of indole. (a) How many \pi electrons does indole have? (b) What is

Amazon.com: Indole Ring Synthesis: From Natural Products to Drug Discovery: 9780470512180: Gribble, Gordon W.: Books