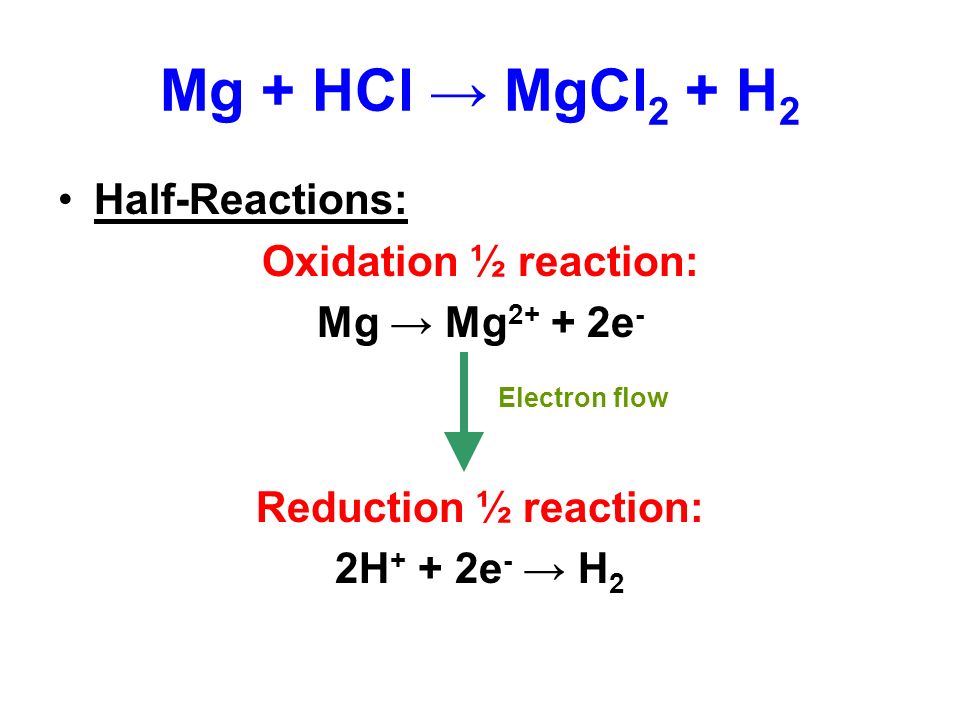
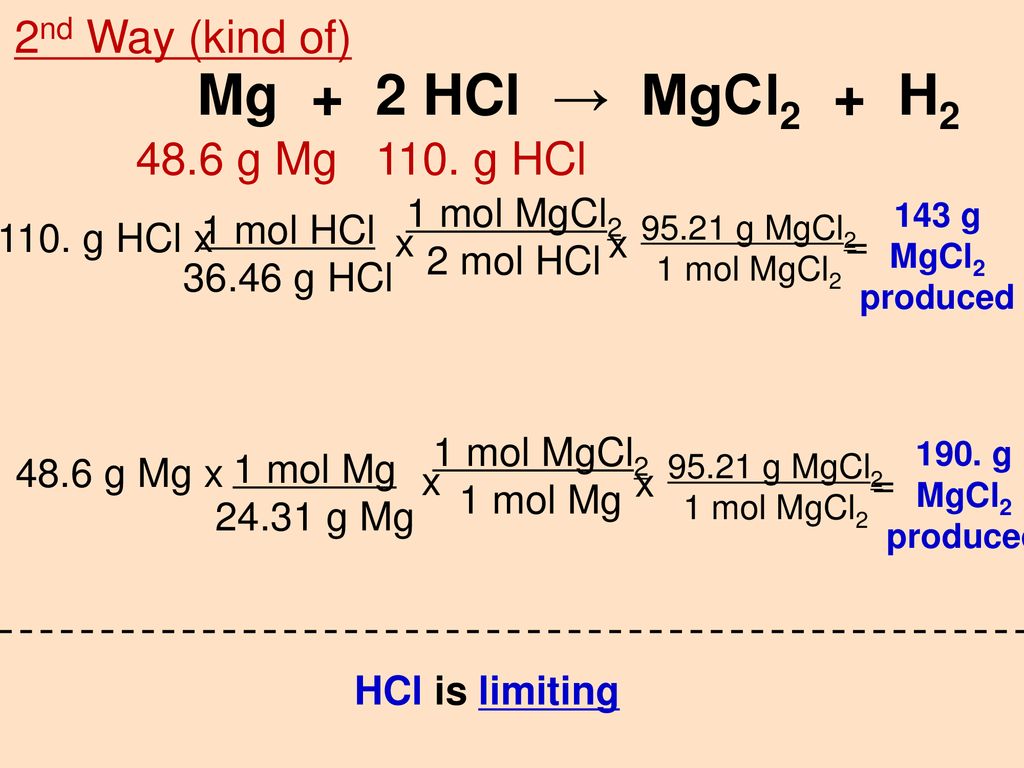
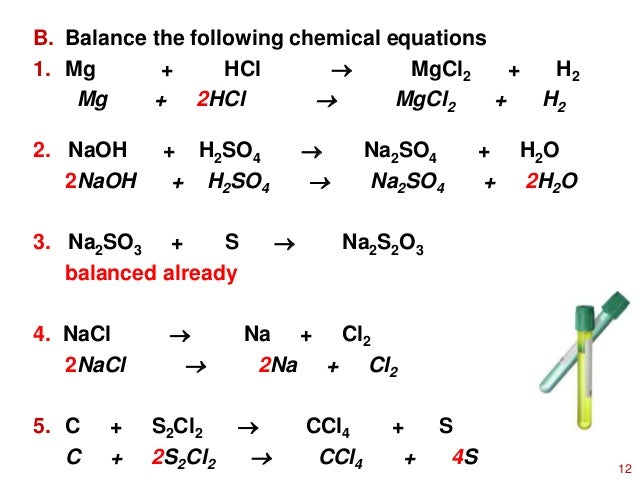
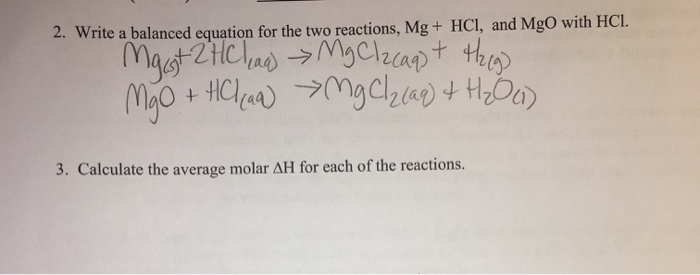SOLVED:Use the balanced chemical equation to determine how many grams of MgCl2 are being produced if you begin with 5.55 g of Mg and the reaction runs with a 60% yield. Mg +

Balance the following equations.(i) KMnO2 + HCl → KCl + MnCl2 + H2 O + Cl2 (ii) NH3 + O3 → NO + H2 O

Mg(OH)2 +HCl =MgCl2 +H2O Balanced Equation||Magnesium Hydroxide +Hydrochloric Acid Balanced Equation - YouTube

Write balanced chemical equations for the following reactions.1. Reaction of sodium with oxygen.2. - Brainly.in

According to the chemical equation, mg(s) + 2hcl mgcl2(aq) + h2(g) how many moles of magnesium are - Brainly.in