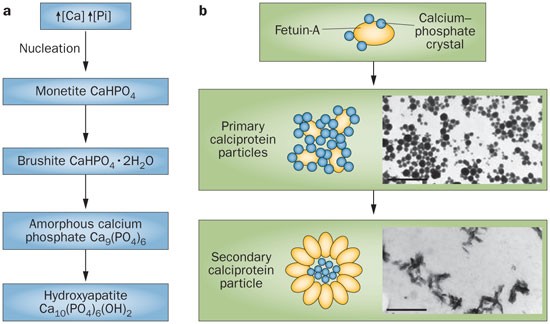
Large differences in phase 3 overall survival spark FDA hold on Oncopeptides' multiple myeloma drug | FierceBiotech

A clinical trial to look at the safety and effectiveness of three different combinations of drugs for treating follicular lymphoma, mosunetuzumab plus lenalidomide, glofitamab plus lenalidomide, or glofitamab plus obinutuzumab and lenalidomide,

A clinical trial to look at the safety and effectiveness of three different combinations of drugs for treating follicular lymphoma, mosunetuzumab plus lenalidomide, glofitamab plus lenalidomide, or glofitamab plus obinutuzumab and lenalidomide,

A clinical trial to look at the safety and effectiveness of three different combinations of drugs for treating follicular lymphoma, mosunetuzumab plus lenalidomide, glofitamab plus lenalidomide, or glofitamab plus obinutuzumab and lenalidomide,