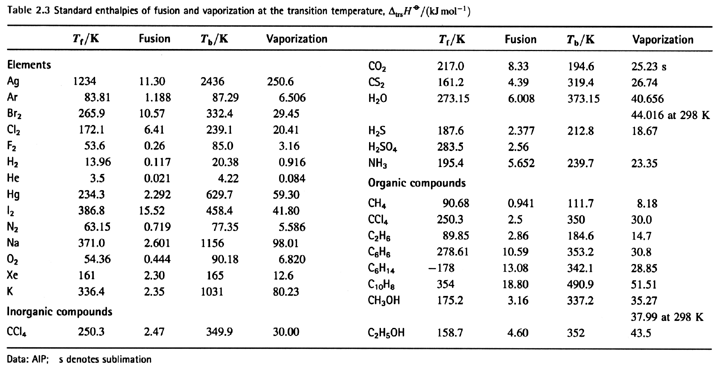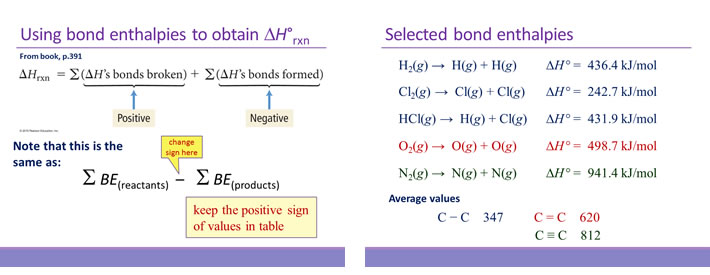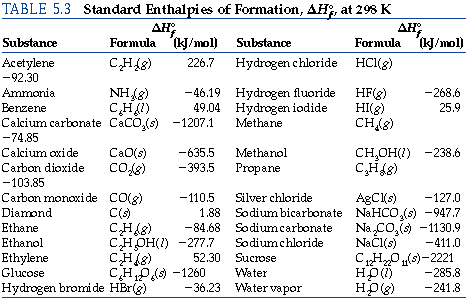
Calculate the enthalpy of reaction for the reaction "CH"_3"COOH" + "H"_2"O" -> "CH"_3"CH"_2"OH" + "O"_2? | Socratic
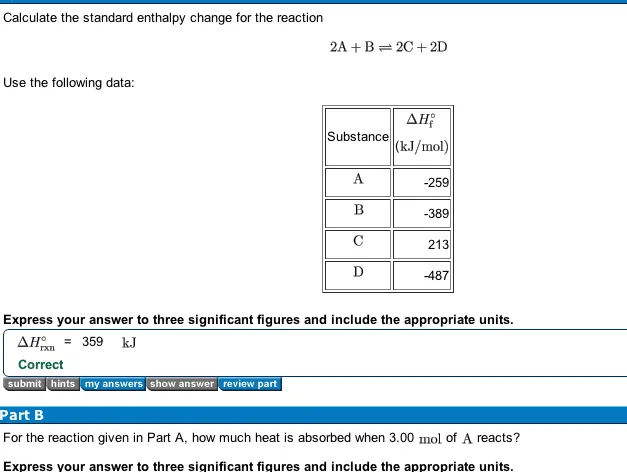
OneClass: Calculate the standard enthalpy change for the reaction 2A + B^2C + 2D Use the following d...

SOLVED:42 What is the overall enthalpy change of the reaction POio(s) _ PaOo(s) + 2 0.(g) given: Pa(s) + } 0z(g) PaOo(s) AH =-1640 kJ P Oi(s) _" Pa(s) 0.(@) AH 2940

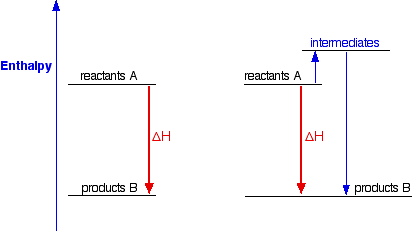
Consider the following enthalpy diagram: mc020-1.jpg Which label shows the overall enthalpy - Brainly.com
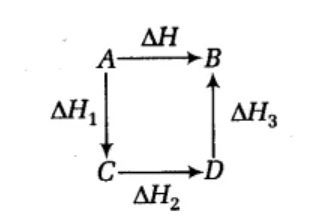
Hess's law states that : ( )A. the standard enthalpy of an overall reaction is the sum of the enthalpy changes in individual reactionsB. enthalpy of formation of a compound is same as the enthalpy of decomposition of the compound into constituent ...