
Sanofi begins work on succession plan; FDA issues warning letters to Lupin's US, India units | Radio Compass Blog

FDA Keeps Brand-Name Drugs On A Fast Path To Market ― Despite Manufacturing Concerns | Kaiser Health News

Teva settles with Roche to launch first Rituxan biosimilar in US - BioProcess InternationalBioProcess International









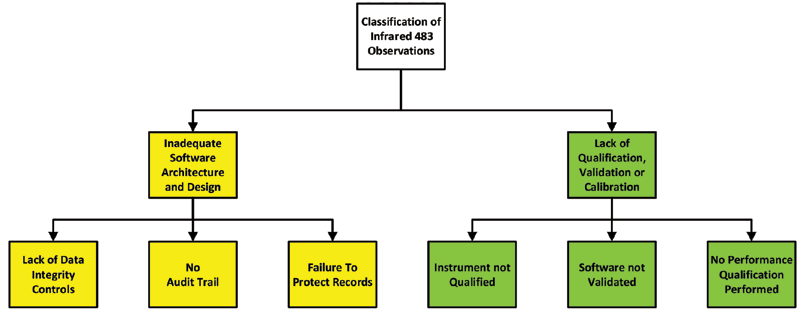







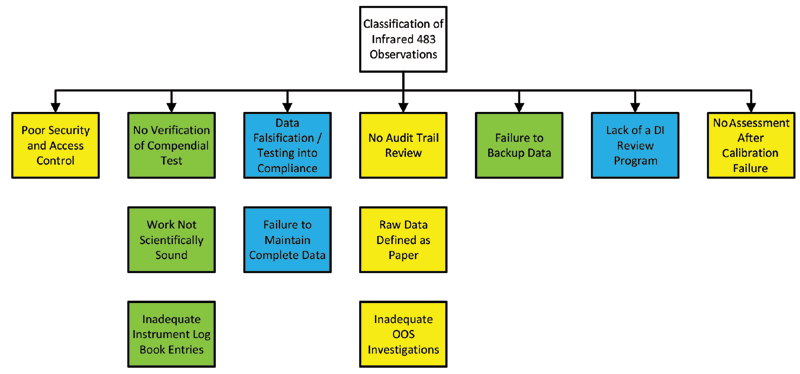



![書籍紹介] FDAのGMP査察から学ぶ 第2版 読めばわかる 査察官の視点・指摘の意図 | PHARM TECH JAPAN ONLINE-製剤技術とGMPの最先端技術情報サイト 書籍紹介] FDAのGMP査察から学ぶ 第2版 読めばわかる 査察官の視点・指摘の意図 | PHARM TECH JAPAN ONLINE-製剤技術とGMPの最先端技術情報サイト](https://jiho-contents.s3-ap-northeast-1.amazonaws.com/s3fs-public/ptj/article/2020/08/FDA%E3%81%AEGMP_23.png?BExJHaC523MfySIolVWjac0.BphKcBZA)